What would be the volume of the balloon that at sea level has a volume of 5 liters when the pressure will drop to p2=0.25 ATM? - Quora

A weather balloon filled with hydrogen at 1 atm and 300 K has volume equal to 12000 liters. On ascending it reaches a place where temperature is 250 K and pressure is

A balloon has a volume of 3.00 liters at 24.0°c. the balloon is heated to 48.0°c. calculate the new volume - Brainly.com
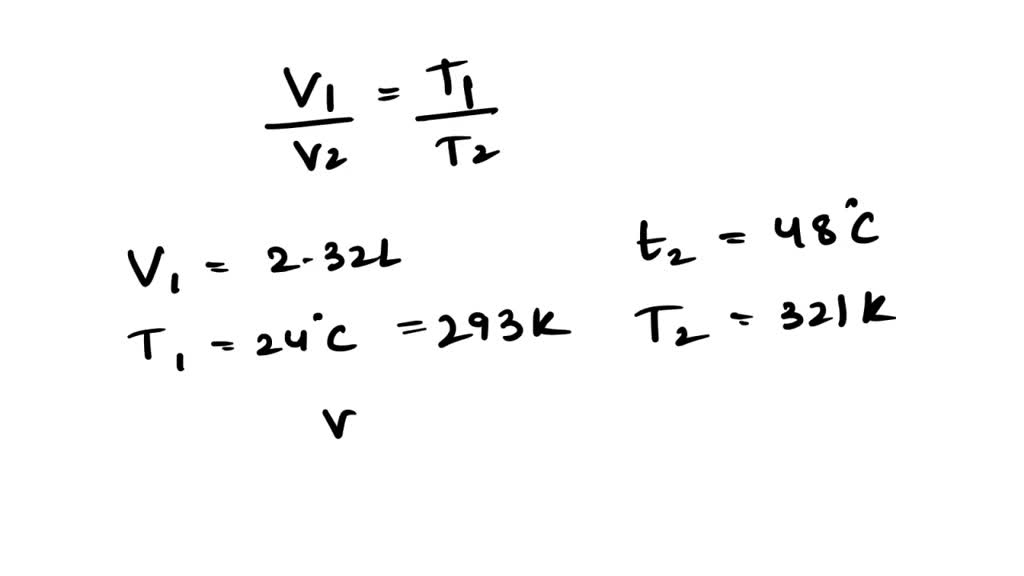
SOLVED: A balloon has a volume of 2.32 liters at 24.0°C. The balloon is heated to 48.0°C. Calculate the new volume of the balloon.

A balloon filled with helium gas at 1.00 atm occupies 11.1 L. What volume would the balloon occupy in the upper atmosphere, at a pressure of 0.25 atm and a constant temperature?

Question Video: Calculating the Moles of a Gas in a Given Volume by Determining the Molar Gas Volume | Nagwa

View question - A Helium filled weather balloon, when released, has a volume of 10.0 L at 27°C and a pressure of 663 mmHg. What Volume, in liters, will the balloon occupy at