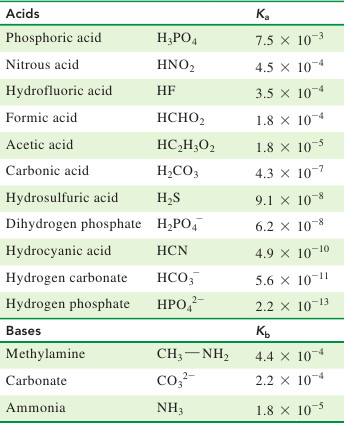
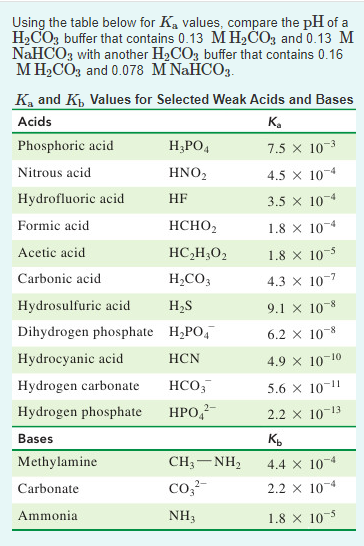
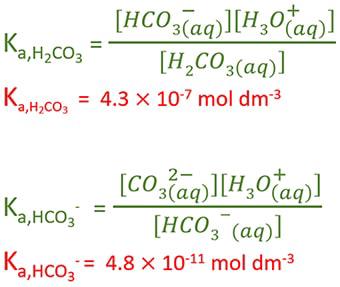
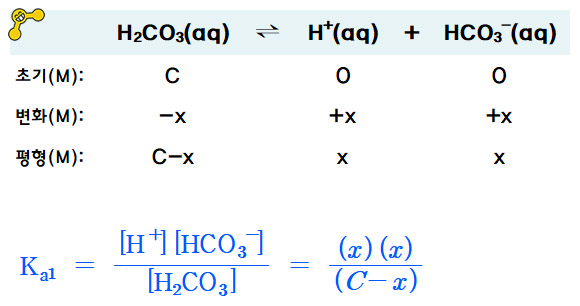SOLVED: Consider a buffer made of carbonic acid HzCO3 and its conjugate base NaHCO3 with a pH of 6.50. (Ka of H2CO3 is 4.3x10-7) [HCO3 (a) Calculate the log needed to produce

Thermal stability of β-H2CO3 in the product mixture at 260 K, ∼400 mbar... | Download Scientific Diagram
Find the concentration of H+, HCO^-3 and CO^2-3, in a 0.01M solution of carbonic acid if the - Sarthaks eConnect | Largest Online Education Community
Why is carbonic acid a weak acid even though it gets completely dissociated into H+ and CO3- ions? - Quora

SOLVED: The blood buffer system is composed of H2CO3 (carbonic acid, Ka = 7.9 x 10^-7 ) and its conjugate base, HCO3- (bicarbonate). In a healthy adult, the pH of blood is

The Ka of carbonic acid is 4.3 x 10-7. H2CO3 = H+ + HCO3 This means that H2co3 is a____. A.good - Brainly.com